We are a Merchant Exporter supplying Chemicals, Pharmaceuticals, Agriculture, Textile products world wide. We have so many products available in Chemicals like Laboratory Chemicals, High purity Solvents, Food Grade Pharmaceuticals, Safety & Hygie...
- (+91) 9998943150
- info@swastiexports.com
- B/84, Shrinivas Tenaments, Behind Makarpura Bus Depot,, , Vadodara, Gujarat, India. 390010
© 2020 Swasti Exports. All Rights Reserved
- Home
- /GC BIOTECH - Expert in Vaccine Adjuvants
GC BIOTECH - Expert in Vaccine Adjuvants
GC Biotech specializes in the research and development of vaccine adjuvants, providing global clients with integrated products, services, and technological solutions. Our scope spans from discovery to pre-clinical and clinical research stages.
Our Goals
- ● Build cost-effective capacity for adjuvant production
- ● Become a reliable partner for human & veterinary vaccines
- ● Design adjuvant systems tailored to specific needs
Our integrated and open-access platform helps reduce R&D costs and accelerate global delivery of new vaccines. We assist in delivering affordable, innovative solutions for infectious diseases, allergies, and cancers.

Technical Support & Solutions
We offer full-spectrum formulation development and manufacturing services, from pre-formulation studies to analytical validation.
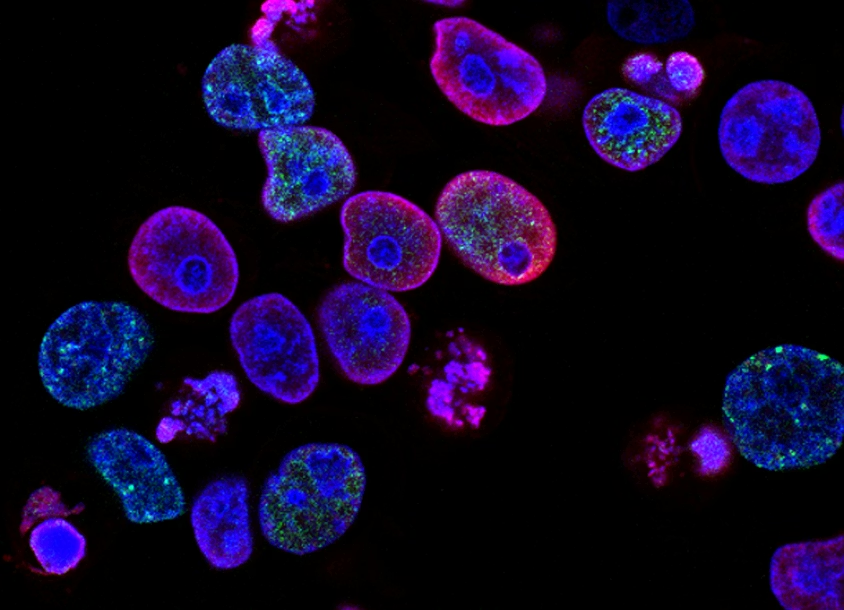
Immune Modulators & Customized Molecules
✓ Multiple options for discovery scales, purification levels, and modification methods meeting GLP/GMP standards.
Delivery Systems & Formulations
Delivery systems for human and veterinary R&D meeting GLP/GMP standards.
Three distinct delivery systems and support for custom immune modulator formulations.
Diverse adjuvant formulations optimized for various vaccine antigens.
Development & Manufacturing
Get Quote
Download cart items as a formal quotation
-
Step 1:
Add items to the Cart
-
Step 2:
Go to My Cart Page
-
Step 3:
Download Quotation


